Recent developments in bioelectronics and power supply miniaturization are providing new design opportunities for in vivo shape memory devices.
Originally featured in AM&P With SMST NewsWire on March 1, 2026.
Is the medical device community on the verge of a new paradigm of in vivo shape memory devices? Much like self-expanding superelastic implantable medical devices revolutionized the industry more than 30 years ago, a new stage is being set. The emergence of miniature leadless pacemakers with sealed power supplies that last multiple years, pulsed field ablation for controlled in vivo heating, and brain computer interfaces with complex bioelectronics all demonstrate the rapidly evolving landscape where implantable power/heat sources are on the verge of becoming commonplace. These advancements in implantable power management may very well usher in a wave of shape memory medical devices that require heat activation.
EARLY MEDICAL-DEVICE DESIGN
Some early medical devices such as the Memotherm Stent and Simon Nitinol IVC Filter were marketed as having shape memory. These devices were tuned with an austenitic finish temperature, Af, above room temperature and below body temperature (e.g., Af = 30 ± 5°C). While these devices feel malleable when handled at room temperature and take on a predefined shape at body temperature, thus the perception by users as having “shape memory,” they more accurately fall under the category of self-expanding superelastic implants. Instead, a true shape memory implant is one in which a predefined shape is achieved through the application of heat while the device is in its target location in the human body (i.e., austenitic start temperature, As, or R-phase start temperature, RS, is > 37°C and the device takes on a new shape when heated above body temperature). With the exception of a few benign prostatic hyperplasia implants that rely upon a warm 55-60°C saline flush to achieve full expansion during implantation (e.g., Memokath, Endocare Horizon Stent), no commercial devices exist that truly utilize the shape memory properties of Nitinol.
A primary obstacle to the use of the shape memory effect in medical devices is the sensitivity of human tissue to damage at relatively low temperatures and short durations. Indeed, tissue damage occurs after just 1 second of exposure to 70°C (Fig. 1). Common binary Nitinol alloy formulations that would seem desirable for in vivo actuation (e.g., Fort Wayne Metals Niti#6 with an As ≥ 35°C) require temperatures as high as 90°C to achieve complete actuation above the Af temperature. Therefore, exceedingly short-duration heating of conventional binary nickel-titanium, or the development of narrower hysteresis ternary/quaternary shape memory alloys may be required for the advancement of shape memory implants.
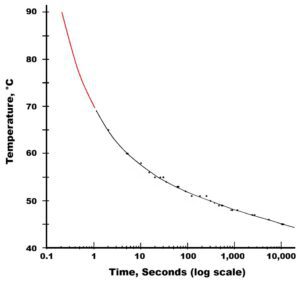
Fig. 1 – Tissue necrosis time versus temperature curve; adapted from Moritz & Henriques with the red portion of the graph extrapolated to 90°C consistent with the Af temperature of common binary Nitinol in vivo shape memory material[1].
NEW PARADIGM
While few commercial implants have used shape memory actuation to date, the aforementioned advancements in implantable power and bioelectronics have inspired a new paradigm. Are these advancements capable of driving the heat-actuation times down to a realm that is safe for surrounding tissue? Three examples of devices currently under development suggest that the answer is “yes.”
Glaucoma is caused by elevated pressures due to fluid buildup in the eye. When pharmaceutical treatments fail, ophthalmologists often turn to implantable shunts to drain fluid from the eye and relieve this pressure (e.g., Ahmed Glaucoma Valve, Baerveldt Glaucoma Implant). Myra Vision’s novel shape memory implant, the CalibrEye System, offers an adjustable fluid outflow that can be tailored to a patient’s dynamic needs as their glaucoma advances. Unlike existing shunts with a fixed drainage flow, the CalibrEye device can double or triple its fluid flow by opening two optional drainage channels that are capped by shape memory Nitinol actuators. Selectively heating the innermost arm of each actuator triggers the cap to open the optional drainage channel, and activating the outermost arm closes the cap (Fig. 2). Atraumatic heating of the Nitinol actuators is achieved using a common green laser system mounted on an ophthalmic slit lamp. Although temperatures of approximately 90°C are required to actuate the device, tissue damage is prevented due to the highly localized spot size (200 µm) that engages only the Nitinol implant, and the incredibly short pulse duration (100 ms).
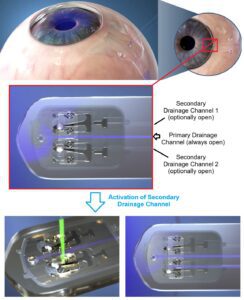
Fig. 2 – The CalibrEye glaucoma shunt provides one primary and two secondary (optional) drainage channels for the management of eye pressure. Two independent thin-film shape memory Nitinol actuators provide caps to the optional channels. When green laser energy activates either of these actuators, the cap moves to an open or closed position to optionally increase or decrease fluid flow from the eye through the secondary drainage channels. Adapted from Chang[2].
Recent innovations suggest that a reduction of heart failure symptoms may be achieved by artificially creating a hole in the atrial septum to shunt blood from the left atrium to the right atrium thereby reducing arterial pressure and fluid load (e.g., Corvia Medical, V-Wave Medical, NoYA Medical, and Adona Medical). Interestingly, two interatrial shunt device companies independently arrived at novel shape memory solutions.
Citing the clinical need that “A flexible shunt size may help meet the specific or changing requirements of individual patients,” Dr. Neal Eigler posed the question “What if a shunt’s orifice could be serially adjusted (larger or smaller) in vivo?”[3] Dr. Eigler then introduced the V-Wave Magical shunt as a solution where the proximal and distal conical anchoring ends of the device remained superelastic while the waist of the shunt underwent a selective thermomechanical treatment to locally tune the Af to ≥ 60°C (Fig. 3). This waist region is manufactured such that the shunt diameter would be smallest when heated above the transition temperature, and deformable at body temperature. In such a configuration, the waist can be enlarged to 9.2 mm via a balloon. To shrink the waist, a warm saline flush actuates the device and reduces the diameter to 4.8 mm. This mechanism of ballooning and saline flushing provides physicians with familiar and established techniques that offer bidirectional movement of a Nitinol device in vivo rather than the unidirectional self-expanding mechanism used in the prior decades.
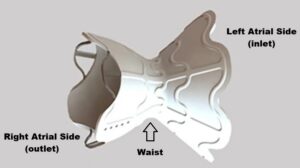
Fig. 3 – V-Wave Ventura device (similar to the Magical device) showing the conical inlet and outlet features of the implant. These superelastic features anchor the device to the patient’s atrial septum. The waist region is locally tuned to render its shape memory property(i.e., As > 37°C). With this pretrained shape, the waist is malleable and expandable via a balloon at body temperature, and can be contracted to its original small diameter when warm saline is flushed through the device.
Developed in parallel, Adona Medical’s Delphi shunt operates on a shape memory principle similar to the V-Wave Magical device. However, unlike the Magical device that uses warm saline, which may risk temperature elevation to adjacent tissue, Adona’s implant uses in vivo induction heating of an electrically and thermally insulated shape memory Nitinol orifice to adjust the shunt diameter without risking a local temperature rise to surrounding tissue[4,5]. Adona’s first-in-human trial successfully demonstrated atraumatic in vivo actuation of their shunt even months after implantation. Importantly, the Adona team envisioned methods whereby bidirectional actuation could be achieved remotely using ex vivo induction heating coils. By nesting two actuators preset to either a small or large size, and by tuning them to respond to different radio frequencies (RF) for heat induction, each of the actuators operate independently to drive the shunt diameter to any desired size by applying the RF signal external to the patient’s chest (Fig. 4).
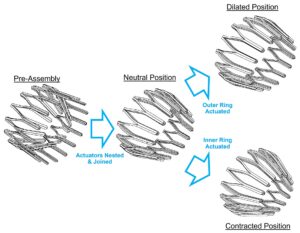
Fig. 4 – Adona Medical’s nested actuator design enables bidirectional actuation within a single device. Two shape memory implants with a preset cylindrical shape and conical shape are thermally isolated from one another, nested, and joined such that they achieve a stable neutral position between a cylindrical and conical shape. When the outer ring (the cylindrical shape) is heated, e.g., by joule heating through an electrical circuit, the composite structure widens to its dilated position, and vice versa when the inner ring (the conical shape) is heated. Adapted from Alexander et al.[6].
It remains unknown if any of the previously mentioned investigational devices will achieve regulatory approval and commercial success. Regardless, they serve to inspire the next generation of medical-device designers to explore shape memory for innovative Nitinol implants.
Early concepts such as removal of implants days/months/years after implantation by shape memory shrinking them back to catheter size or articulated robotic arms for remote surgery that have since been abandoned in favor of mechanical pull-wire mechanisms may now be worth revisiting in the modern era[7,8]. Arguably, the most impactful opportunity for shape memory innovation is in the field of pediatric devices where the patient’s size (and hence target therapeutic size of the implant) changes substantially throughout their lifetime. Are these size modifications achievable through unique shape memory actuator designs that eliminate traumatic surgeries currently performed to remove obsolete devices and replace them with upsized implants as the child matures to adulthood?
CHALLENGES AND OPPORTUNITIES
Several challenges (i.e., opportunities for innovation) lie ahead for designers wishing to use shape memory for implantable devices:
- Can binary Nitinol, with its established biocompatibility and regulatory pathway, provide the necessary actuation movements and/or forces using times and temperatures below which would traumatize human tissue?
- If ternary or quaternary alloys are pursued to overcome temperature limitations (e.g., NiTiCu with its very narrow temperature hysteresis) what are the biocompatibility and consequent regulatory challenges for those alloys that haven’t historically been proven in vivo?
- Manufacturing techniques must be developed to selectively tailor regions of a device to be shape memory while retaining regions of superelasticity elsewhere in the same implant such as that developed by the V-Wave Medical team.
- What methods of heat delivery (e.g., warm saline, induction heating, joule heating) will be available to safely deliver the power required to trigger in-vivo actuation?
Self-expandable superelastic devices have evolved from the humble beginnings of simple wire-form stents into the complex laser-cut metal/polymer/tissue composite heart valves that are commonplace today. Having been involved in the emerging field of shape memory implants and given the recent advancements in bioelectronics as well as power supply miniaturization, this author is optimistic that the industry is at a similar entry point of exponential growth for in vivo actuatable shape memory devices that are beyond current imagination.
Note: The V-Wave Magical and the V-Wave Ventura are registered trademarks of Johnson & Johnson.
For more information: Scott Robertson, Vice President of Nitinol Technology, scottrobertson@resonetics.com
REFERENCES
- R. Moritz and FC Henriques, Jr., The Relative Importance of Time and Surface Temperature in the Causation of Cutaneous Burns, The American Journal of Pathology, 23(5) p 695-720, September 1947.
- Chang, Myra Vision CalibEye System, Ophthamology Innovation Summit 2023, slides and presentation accessed 1.8.2026 at www.youtube.com/watch?v=yuurRcIPgHY.
- Eigler, Precision Interatrial Shunting…Matching the Device to the Patient, Innovations in Cardiovascular Interventions meeting; December 6, 2022.
- Hammerand, Shunt Device Startup Wants to Break Barriers in Nitinol and Heart Sensing, Medical Design & Outsourcing, August 15, 2023, www.medicaldesignandoutsourcing.com/adona-medical-adjustable-heart-shunt-nitinol-biatrial-pressure-sensing.
- Hammerand, This Shifamed Startup Shares the Shape Memory Secret Behind its Nitinol Heart Failure Implant, Medical Design & Outsourcing, December 1, 2025, www.medicaldesignandoutsourcing.com/adona-medical-shape-memory-nitinol-heart-failure-adjustable-shunt.
- Alexander, S. Robertson, et al., Shape Memory Actuators for Adjustable Shunting Systems, and Associated Systems and Methods, US 12,090,290 B2, United States Patent and Trademark Office, September 17, 2024.
- E. Bramfitt and R.L. Hess, A Novel Heat Activated Recoverable Temporary Stent (HARTS System); Shape Memory and Superelastic Technologies Conference 1994.
- Dario, M.C. Montesi, Shape Memory Alloy Microactuators for Minimal Invasive Surgery, Shape Memory and Superelastic Technologies Conference 1994.